Jump to navigation
Jump to search
NCBI: 03-AUG-2016
⊟Summary[edit | edit source]
- organism: Staphylococcus aureus NCTC8325
- locus tag: SAOUHSC_01105
- pan locus tag?: SAUPAN003389000
- symbol: sdhB
- pan gene symbol?: sdhB
- synonym:
- product: succinate dehydrogenase iron-sulfur subunit
⊟Genome View[edit | edit source]
⊟Gene[edit | edit source]
⊟General[edit | edit source]
- type: CDS
- locus tag: SAOUHSC_01105
- symbol: sdhB
- product: succinate dehydrogenase iron-sulfur subunit
- replicon: chromosome
- strand: +
- coordinates: 1067102..1067917
- length: 816
- essential: no DEG other strains
⊟Accession numbers[edit | edit source]
- Gene ID: 3920747 NCBI
- RefSeq: YP_499649 NCBI
- BioCyc: G1I0R-1038 BioCyc
- MicrobesOnline: 1289562 MicrobesOnline
⊟Phenotype[edit | edit source]
Share your knowledge and add information here. [edit]
⊟DNA sequence[edit | edit source]
- 1
61
121
181
241
301
361
421
481
541
601
661
721
781ATGACTGAACAATCAGTGAAAAACACTCCACAACATGAAACACAATCTAAACCGAAACAA
AAAACAGTAAAATTAATTATTAAACGACAAGATACAAGTGATTCTAAGCCTTATGAAGAA
ACATTTGAAATTCCATATCGTGAAAATTTAAACGTCATTGCTTGTTTAATGGAAATTAGA
CGTAACCCAGTTAATATTAAAGGTGAAAAAACAACACCTGTTGTCTGGGATATGAACTGC
TTAGAAGAAGTATGTGGAGCATGTTCTATGGTTATCAATGGTCGTGCAAGACAATCTTGT
TCTGCGATTGTTGATCAATTAGAACAACCTATTCGTTTAGAGCCAATGAATACTTTCCCA
GTTATCCGTGACTTACAAGTTGATCGTTCTAGAATGTTCGATAACTTAAAACGTATGAAA
GCATGGATCCCAATTGATGGAACGTATGATTTAGGTCCGGGACCACGTATGCCAGAGAAA
AAACGTCAAACAGCTTATGAATTATCTAAATGTATGACATGTGGTGTATGTTTAGAGGTT
TGTCCTAATGTTACTGAAAATAATAAATTCGTTGGTGCACAAGCAATCTCGCAAGTTCGT
TTGTTTAATTTGCACCCAACAGGATCTATGACTAAAGATGAACGTTTAAATGCATTAATG
GGTACTGGTGGCTTACAGCAGTGTGGTAATTCACAAAACTGTGTTAATGCTTGCCCTAAA
GGTATTCCATTAACAACATCCATTGCAGCAATGAACAGAGAAACAACATTCCACATGTTT
AAATCATTCTTTGGTTCAGACCATGAAGTAGAATAA60
120
180
240
300
360
420
480
540
600
660
720
780
816
⊟Protein[edit | edit source]
⊟General[edit | edit source]
- locus tag: SAOUHSC_01105
- symbol: SdhB
- description: succinate dehydrogenase iron-sulfur subunit
- length: 271
- theoretical pI: 7.99685
- theoretical MW: 30583.1
- GRAVY: -0.501845
⊟Function[edit | edit source]
- reaction: EC 1.3.99.1? ExPASyDeleted entry
- TIGRFAM: Energy metabolism TCA cycle succinate dehydrogenase and fumarate reductase iron-sulfur protein (TIGR00384; HMM-score: 137.1)Energy metabolism Anaerobic succinate dehydrogenase and fumarate reductase iron-sulfur protein (TIGR00384; HMM-score: 137.1)Energy metabolism Aerobic succinate dehydrogenase and fumarate reductase iron-sulfur protein (TIGR00384; HMM-score: 137.1)and 25 moreEnergy metabolism Electron transport NADH-quinone oxidoreductase, chain I (TIGR01971; HMM-score: 19)Unknown function General archaeoflavoprotein, MJ0208 family (TIGR02700; HMM-score: 18.7)cytochrome c oxidase accessory protein CcoG (TIGR02745; HMM-score: 18)CoB--CoM heterodisulfide reductase, subunit C (TIGR03290; EC 1.8.98.1; HMM-score: 15.4)4-hydroxybenzoyl-CoA reductase, gamma subunit (TIGR03193; EC 1.3.7.9; HMM-score: 15.1)Purines, pyrimidines, nucleosides, and nucleotides Other selenium-dependent xanthine dehydrogenase (TIGR03311; EC 1.17.1.4; HMM-score: 15.1)pyruvate:ferredoxin (flavodoxin) oxidoreductase (TIGR02176; EC 1.2.7.1; HMM-score: 14.1)Energy metabolism Amino acids and amines choline TMA-lyase-activating enzyme (TIGR04395; EC 1.97.-.-; HMM-score: 13.8)Energy metabolism Anaerobic glycerol-3-phosphate dehydrogenase, anaerobic, C subunit (TIGR03379; EC 1.1.5.3; HMM-score: 13.5)2-oxoacid:acceptor oxidoreductase, delta subunit, pyruvate/2-ketoisovalerate family (TIGR02179; HMM-score: 13.2)Energy metabolism Electron transport NADH-plastoquinone oxidoreductase, I subunit (TIGR00403; EC 1.6.5.3; HMM-score: 13.1)Energy metabolism Electron transport iron-sulfur cluster-binding protein (TIGR00273; HMM-score: 12)Energy metabolism Methanogenesis putative methanogenesis marker 16 metalloprotein (TIGR03287; HMM-score: 11.9)Protein synthesis tRNA and rRNA base modification epoxyqueuosine reductase (TIGR00276; EC 1.-.-.-; HMM-score: 11.8)Energy metabolism Electron transport cytochrome c nitrite reductase, Fe-S protein (TIGR03149; EC 1.7.2.2; HMM-score: 11.5)Energy metabolism Electron transport ferredoxin-type protein NapF (TIGR00402; HMM-score: 10.3)[FeFe] hydrogenase, group A (TIGR02512; EC 1.12.-.-; HMM-score: 9.9)Energy metabolism Chemoautotrophy CO dehydrogenase/acetyl-CoA synthase complex, epsilon subunit (TIGR00314; EC 1.2.99.2; HMM-score: 9.4)methylamine methyltransferase corrinoid protein reductive activase (TIGR04270; HMM-score: 9.4)Central intermediary metabolism Nitrogen fixation ferredoxin III, nif-specific (TIGR02936; HMM-score: 8.2)Energy metabolism Electron transport electron transport complex, RnfABCDGE type, C subunit (TIGR01945; HMM-score: 7.7)glycyl-radical enzyme activating protein (TIGR02494; EC 1.97.1.-; HMM-score: 6.8)Protein fate Protein modification and repair glycine radical enzyme activase, YjjW family (TIGR04041; EC 1.97.1.-; HMM-score: 6.6)ferredoxin-type protein, NapH/MauN family (TIGR02163; HMM-score: 6.5)Central intermediary metabolism Sulfur metabolism sulfite reductase, dissimilatory-type beta subunit (TIGR02066; EC 1.8.99.3; HMM-score: 5.3)
- TheSEED :
- Succinate dehydrogenase iron-sulfur protein (EC 1.3.99.1)
Carbohydrates Central carbohydrate metabolism TCA Cycle Succinate dehydrogenase iron-sulfur protein (EC 1.3.99.1)and 2 more - PFAM: Fer2 (CL0486) Fer2_3; 2Fe-2S iron-sulfur cluster binding domain (PF13085; HMM-score: 100.3)and 17 more4Fe-4S (CL0344) Fer4_8; 4Fe-4S dicluster domain (PF13183; HMM-score: 36.2)Fer4_7; 4Fe-4S dicluster domain (PF12838; HMM-score: 34.3)Fer4_9; 4Fe-4S dicluster domain (PF13187; HMM-score: 30.7)Fer4_10; 4Fe-4S dicluster domain (PF13237; HMM-score: 26.9)Fer4_17; 4Fe-4S dicluster domain (PF13534; HMM-score: 24.6)Fer4_2; 4Fe-4S binding domain (PF12797; HMM-score: 19.5)Fer4_16; 4Fe-4S double cluster binding domain (PF13484; HMM-score: 19.5)Fer4_13; 4Fe-4S single cluster domain of Ferredoxin I (PF13370; HMM-score: 19.2)Fer4; 4Fe-4S binding domain (PF00037; HMM-score: 17.8)Fer4_21; 4Fe-4S dicluster domain (PF14697; HMM-score: 15.1)Fer4_15; 4Fe-4S single cluster domain (PF13459; HMM-score: 15)Fer4_4; 4Fe-4S binding domain (PF12800; HMM-score: 14.1)no clan defined P9_TM; Minor capsid protein P9 transmembrane helices (PF19066; HMM-score: 14)4Fe-4S (CL0344) Fer4_3; 4Fe-4S binding domain (PF12798; HMM-score: 12.6)Fer4_18; 4Fe-4S dicluster domain (PF13746; HMM-score: 12.6)Fer4_6; 4Fe-4S binding domain (PF12837; HMM-score: 11.9)Fer4_22; 4Fe-4S dicluster domain (PF17179; HMM-score: 7.6)
⊟Structure, modifications & cofactors[edit | edit source]
- domains:
- modifications:
- cofactors:
- effectors:
⊟Localization[edit | edit source]
- PSORTb: Cytoplasmic
- Cytoplasmic Score: 9.89
- Cytoplasmic Membrane Score: 0.09
- Cellwall Score: 0.01
- Extracellular Score: 0.02
- Internal Helices: 0
- DeepLocPro: Cytoplasmic Membrane
- Cytoplasmic Score: 0.3113
- Cytoplasmic Membrane Score: 0.6484
- Cell wall & surface Score: 0
- Extracellular Score: 0.0403
- LocateP: Intracellular
- Prediction by SwissProt Classification: Cytoplasmic
- Pathway Prediction: No pathway
- Intracellular possibility: 1
- Signal peptide possibility: -1
- N-terminally Anchored Score: 1
- Predicted Cleavage Site: No CleavageSite
- SignalP: no predicted signal peptide
- SP(Sec/SPI): 0.004685
- TAT(Tat/SPI): 0.00098
- LIPO(Sec/SPII): 0.000969
- predicted transmembrane helices (TMHMM): 0
⊟Accession numbers[edit | edit source]
⊟Protein sequence[edit | edit source]
- MTEQSVKNTPQHETQSKPKQKTVKLIIKRQDTSDSKPYEETFEIPYRENLNVIACLMEIRRNPVNIKGEKTTPVVWDMNCLEEVCGACSMVINGRARQSCSAIVDQLEQPIRLEPMNTFPVIRDLQVDRSRMFDNLKRMKAWIPIDGTYDLGPGPRMPEKKRQTAYELSKCMTCGVCLEVCPNVTENNKFVGAQAISQVRLFNLHPTGSMTKDERLNALMGTGGLQQCGNSQNCVNACPKGIPLTTSIAAMNRETTFHMFKSFFGSDHEVE
⊟Experimental data[edit | edit source]
- experimentally validated: PeptideAtlas [1] [2]
- protein localization: data available for COL
- quantitative data / protein copy number per cell:
- interaction partners:
⊟Expression & Regulation[edit | edit source]
⊟Operon[edit | edit source]
- MicrobesOnline: SAOUHSC_01103 > sdhA > sdhBpredicted SigA promoter [3] : S455 > SAOUHSC_01103 > sdhA > sdhB > S456 > SAOUHSC_01106 > SAOUHSC_01107 > SAOUHSC_01108predicted SigB promoter [3] : S455 > SAOUHSC_01103 > sdhA > sdhB > S456 > SAOUHSC_01106 > SAOUHSC_01107 > SAOUHSC_01108
⊟Regulation[edit | edit source]
- regulator: CcpA* regulon
CcpA* (TF) important in Carbon catabolism; RegPrecise transcription unit transferred from N315 data RegPrecise
⊟Transcription pattern[edit | edit source]
- S.aureus Expression Data Browser: [3]
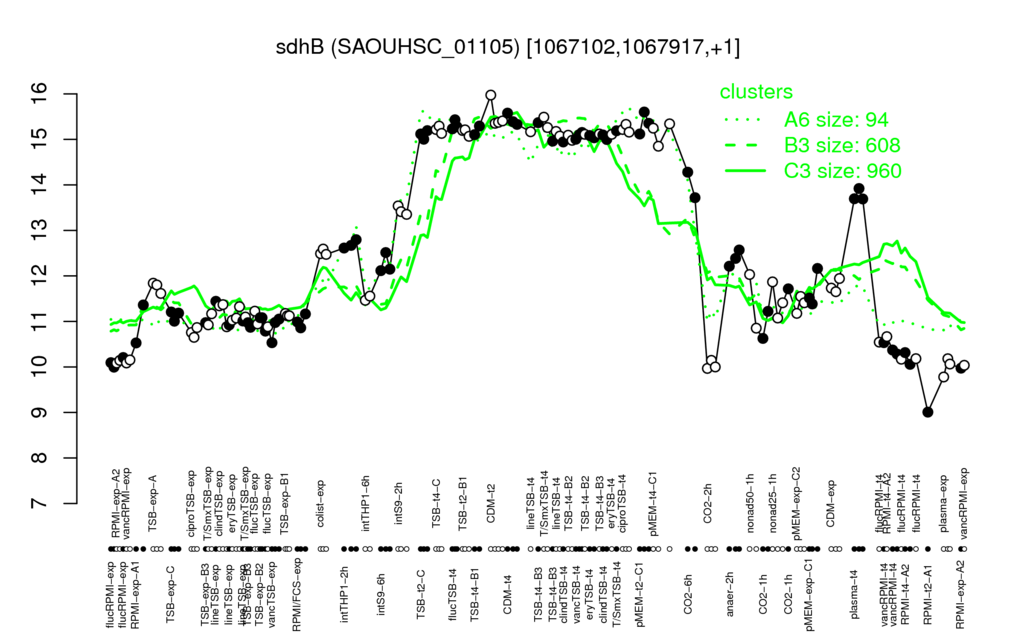 Multi-gene expression profiles
Multi-gene expression profiles
⊟Protein synthesis (provided by Aureolib)[edit | edit source]
- Aureolib: no data available
⊟Protein stability[edit | edit source]
- half-life: no data available
⊟Biological Material[edit | edit source]
⊟Mutants[edit | edit source]
⊟Expression vector[edit | edit source]
⊟lacZ fusion[edit | edit source]
⊟GFP fusion[edit | edit source]
⊟two-hybrid system[edit | edit source]
⊟FLAG-tag construct[edit | edit source]
⊟Antibody[edit | edit source]
⊟Other Information[edit | edit source]
You can add further information about the gene and protein here. [edit]
⊟Literature[edit | edit source]
⊟References[edit | edit source]
- ↑ Maren Depke, Stephan Michalik, Alexander Rabe, Kristin Surmann, Lars Brinkmann, Nico Jehmlich, Jörg Bernhardt, Michael Hecker, Bernd Wollscheid, Zhi Sun, Robert L Moritz, Uwe Völker, Frank Schmidt
A peptide resource for the analysis of Staphylococcus aureus in host-pathogen interaction studies.
Proteomics: 2015, 15(21);3648-61
[PubMed:26224020] [WorldCat.org] [DOI] (I p) - ↑ Stephan Michalik, Maren Depke, Annette Murr, Manuela Gesell Salazar, Ulrike Kusebauch, Zhi Sun, Tanja C Meyer, Kristin Surmann, Henrike Pförtner, Petra Hildebrandt, Stefan Weiss, Laura Marcela Palma Medina, Melanie Gutjahr, Elke Hammer, Dörte Becher, Thomas Pribyl, Sven Hammerschmidt, Eric W Deutsch, Samuel L Bader, Michael Hecker, Robert L Moritz, Ulrike Mäder, Uwe Völker, Frank Schmidt
A global Staphylococcus aureus proteome resource applied to the in vivo characterization of host-pathogen interactions.
Sci Rep: 2017, 7(1);9718
[PubMed:28887440] [WorldCat.org] [DOI] (I e) - ↑ 3.0 3.1 3.2 Ulrike Mäder, Pierre Nicolas, Maren Depke, Jan Pané-Farré, Michel Debarbouille, Magdalena M van der Kooi-Pol, Cyprien Guérin, Sandra Dérozier, Aurelia Hiron, Hanne Jarmer, Aurélie Leduc, Stephan Michalik, Ewoud Reilman, Marc Schaffer, Frank Schmidt, Philippe Bessières, Philippe Noirot, Michael Hecker, Tarek Msadek, Uwe Völker, Jan Maarten van Dijl
Staphylococcus aureus Transcriptome Architecture: From Laboratory to Infection-Mimicking Conditions.
PLoS Genet: 2016, 12(4);e1005962
[PubMed:27035918] [WorldCat.org] [DOI] (I e)
⊟Relevant publications[edit | edit source]
Rosmarie Gaupp, Steffen Schlag, Manuel Liebeke, Michael Lalk, Friedrich Götz
Advantage of upregulation of succinate dehydrogenase in Staphylococcus aureus biofilms.
J Bacteriol: 2010, 192(9);2385-94
[PubMed:20207757] [WorldCat.org] [DOI] (I p)
