Jump to navigation
Jump to search
NCBI: 03-AUG-2016
⊟Summary[edit | edit source]
- organism: Staphylococcus aureus NCTC8325
- locus tag: SAOUHSC_01269
- pan locus tag?: SAUPAN003596000
- symbol: SAOUHSC_01269
- pan gene symbol?: miaB
- synonym:
- product: (dimethylallyl)adenosine tRNA methylthiotransferase
⊟Genome View[edit | edit source]
⊟Gene[edit | edit source]
⊟General[edit | edit source]
- type: CDS
- locus tag: SAOUHSC_01269
- symbol: SAOUHSC_01269
- product: (dimethylallyl)adenosine tRNA methylthiotransferase
- replicon: chromosome
- strand: +
- coordinates: 1223950..1225494
- length: 1545
- essential: no DEG other strains
⊟Accession numbers[edit | edit source]
- Gene ID: 3919922 NCBI
- RefSeq: YP_499802 NCBI
- BioCyc: G1I0R-1187 BioCyc
- MicrobesOnline: 1289716 MicrobesOnline
⊟Phenotype[edit | edit source]
Share your knowledge and add information here. [edit]
⊟DNA sequence[edit | edit source]
- 1
61
121
181
241
301
361
421
481
541
601
661
721
781
841
901
961
1021
1081
1141
1201
1261
1321
1381
1441
1501GTGAACGAAGAACAAAGAAAAGCAAGTTCTGTAGATGTTTTAGCTGAGAGAGATAAGAAA
GCAGAAAAAGATTATAGTAAATATTTTGAACATGTTTATCAGCCGCCTAATTTAAAAGAA
GCGAAAAAAAGAGGTAAACAAGAAGTTCGTTATAATAGAGATTTCCAAATTGATGAAAAA
TATCGCGGTATGGGGAACGAGCGTACATTTTTAATTAAAACATATGGATGTCAAATGAAT
GCACATGACACTGAGGTCATTGCTGGTATACTTGAAGCATTAGGCTATCAAGCAACGACT
GATATTAACACTGCAGATGTTATTTTAATTAATACATGTGCGATTAGAGAAAATGCCGAG
AACAAAGTGTTTAGTGAAATAGGTAATTTGAAGCATTTGAAAAAAGAACGACCTGATATT
TTAATCGGTGTTTGTGGTTGTATGTCACAAGAAGAGTCAGTAGTGAATAAAATTTTAAAA
TCGTATCAAAATGTAGATATGATATTTGGTACACATAATATTCATCATTTACCAGAAATT
TTAGAAGAAGCATACTTATCTAAAGCAATGGTTGTTGAAGTATGGTCTAAAGAAGGAGAC
GTTATTGAAAATCTTCCAAAAGTCCGTGAAGGCAACATTAAAGCATGGGTCAATATTATG
TATGGTTGTGATAAGTTTTGTACATATTGTATTGTTCCATTTACAAGAGGTAAAGAACGA
AGCCGTAGACCTGAAGACATTATAGATGAAGTACGTGAACTTGCTCGTGAAGGTTACAAA
GAAATAACGCTTTTAGGTCAAAATGTAAATTCTTATGGTAAAGATTTACAGGATATAGAA
TATGACTTAGGAGATCTTTTACAAGCAATTTCTAAAATAGCGATTCCAAGAGTTCGTTTC
ACAACAAGTCATCCTTGGGACTTTACAGATCACATGATTGATGTTATTTCAGAGGGTGGT
AATATCGTTCCGCATATCCACTTGCCAGTTCAATCTGGAAATAATGCAGTATTAAAAATA
ATGGGTAGAAAATATACACGAGAAAGTTATTTGGATTTAGTAAAACGAATCAAAGATAGA
ATTCCTAATGTAGCATTAACTACAGATATTATTGTAGGGTATCCAAATGAATCAGAGGAA
CAATTTGAAGAAACTTTAACTCTGTATGATGAAGTTGGTTTTGAACATGCATATACGTAC
TTGTATTCACAACGTGATGGTACGCCTGCTGCTAAAATGAAAGATAATGTACCTTTAAAT
GTCAAAAAGGAACGATTGCAACGTTTGAATAAAAAAGTTGGTCATTATTCACAAATAGCT
ATGAGTAAGTACGAAGGACAAACTGTAACAGTACTTTGTGAAGGAAGTAGTAAAAAAGAT
GATCAGGTTCTTGCTGGCTACACTGATAAAAATAAGCTAGTTAATTTCAAAGCGCCTAAA
GAAATGATTGGTAAACTAGTGGAAGTACGAATAGATGAAGCTAAACAGTATTCATTAAAT
GGCAGTTTTATAAAGGAAGTAGAGCCGGAAATGGTGATTCAATAA60
120
180
240
300
360
420
480
540
600
660
720
780
840
900
960
1020
1080
1140
1200
1260
1320
1380
1440
1500
1545
⊟Protein[edit | edit source]
⊟General[edit | edit source]
- locus tag: SAOUHSC_01269
- symbol: SAOUHSC_01269
- description: (dimethylallyl)adenosine tRNA methylthiotransferase
- length: 514
- theoretical pI: 6.26459
- theoretical MW: 58930.8
- GRAVY: -0.545914
⊟Function[edit | edit source]
- reaction: EC 2.8.4.3? ExPASytRNA-2-methylthio-N6-dimethylallyladenosine synthase N6-dimethylallyladenine37 in tRNA + sulfur-(sulfur carrier) + 2 S-adenosyl-L-methionine + reduced electron acceptor = 2-methylthio-N6-dimethylallyladenine37 in tRNA + S-adenosyl-L-homocysteine + (sulfur carrier) + L-methionine + 5'-deoxyadenosine + electron acceptor
- TIGRFAM: Protein synthesis tRNA and rRNA base modification tRNA-i(6)A37 thiotransferase enzyme MiaB (TIGR01574; EC 2.-.-.-; HMM-score: 630.8)radical SAM methylthiotransferase, MiaB/RimO family (TIGR00089; EC 2.1.1.-,2.8.1.-; HMM-score: 528)and 16 moreProtein synthesis tRNA and rRNA base modification MiaB-like tRNA modifying enzyme (TIGR01579; HMM-score: 334.6)Protein synthesis Ribosomal proteins: synthesis and modification ribosomal protein S12 methylthiotransferase RimO (TIGR01125; EC 2.1.1.-,2.8.1.-; HMM-score: 284.3)Protein synthesis tRNA and rRNA base modification MiaB-like tRNA modifying enzyme, archaeal-type (TIGR01578; HMM-score: 261.7)Unknown function Enzymes of unknown specificity B12-binding domain/radical SAM domain protein, MJ_0865 family (TIGR04014; HMM-score: 82.7)Unknown function Enzymes of unknown specificity B12-binding domain/radical SAM domain protein, MJ_1487 family (TIGR04013; HMM-score: 63.1)Cellular processes Toxin production and resistance radical SAM P-methyltransferase, PhpK family (TIGR04479; EC 2.1.-.-; HMM-score: 40)hopanoid biosynthesis associated radical SAM protein HpnJ (TIGR03471; HMM-score: 29.5)putative heme utilization radical SAM enzyme HutW (TIGR04107; HMM-score: 29.3)Biosynthesis of cofactors, prosthetic groups, and carriers Heme, porphyrin, and cobalamin coproporphyrinogen dehydrogenase HemZ (TIGR03994; EC 1.3.99.22; HMM-score: 22.7)Unknown function Enzymes of unknown specificity radical SAM protein, TIGR01212 family (TIGR01212; HMM-score: 22.6)Biosynthesis of cofactors, prosthetic groups, and carriers Heme, porphyrin, and cobalamin putative oxygen-independent coproporphyrinogen III oxidase (TIGR00539; EC 1.3.99.22; HMM-score: 15.4)putative variant cofactor biosynthesis B12-binding domain/radical SAM domain protein 1 (TIGR04385; HMM-score: 14.2)Biosynthesis of cofactors, prosthetic groups, and carriers Heme, porphyrin, and cobalamin oxygen-independent coproporphyrinogen III oxidase (TIGR00538; EC 1.3.99.22; HMM-score: 13.9)Cellular processes Toxin production and resistance tryptophan 2-C-methyltransferase (TIGR04428; EC 2.1.1.106; HMM-score: 13.2)Unknown function Enzymes of unknown specificity uncharacterized radical SAM protein YgiQ (TIGR03904; HMM-score: 12.9)magnesium-protoporphyrin IX monomethyl ester anaerobic oxidative cyclase (TIGR02026; HMM-score: 11.7)
- TheSEED :
- tRNA-i(6)A37 methylthiotransferase
Protein Metabolism Protein processing and modification Ribosomal protein S12p Asp methylthiotransferase tRNA-i(6)A37 methylthiotransferaseand 3 moreRNA Metabolism RNA processing and modification Methylthiotransferases tRNA-i(6)A37 methylthiotransferase - PFAM: TIM_barrel (CL0036) Radical_SAM; Radical SAM superfamily (PF04055; HMM-score: 118.2)CheY (CL0304) UPF0004; Uncharacterized protein family UPF0004 (PF00919; HMM-score: 106.4)and 2 moreOB (CL0021) TRAM; TRAM domain (PF01938; HMM-score: 46)TRAM_2; TRAM domain (PF18693; HMM-score: 19.4)
⊟Structure, modifications & cofactors[edit | edit source]
- domains:
- modifications:
- cofactors: [4Fe-4S] cluster
- effectors:
⊟Localization[edit | edit source]
- PSORTb: Cytoplasmic
- Cytoplasmic Score: 7.5
- Cytoplasmic Membrane Score: 1.15
- Cellwall Score: 0.62
- Extracellular Score: 0.73
- Internal Helices: 0
- DeepLocPro: Cytoplasmic
- Cytoplasmic Score: 0.9607
- Cytoplasmic Membrane Score: 0.0144
- Cell wall & surface Score: 0.0002
- Extracellular Score: 0.0247
- LocateP: Intracellular
- Prediction by SwissProt Classification: Cytoplasmic
- Pathway Prediction: No pathway
- Intracellular possibility: 1
- Signal peptide possibility: -1
- N-terminally Anchored Score: 1
- Predicted Cleavage Site: No CleavageSite
- SignalP: no predicted signal peptide
- SP(Sec/SPI): 0.009083
- TAT(Tat/SPI): 0.002069
- LIPO(Sec/SPII): 0.003372
- predicted transmembrane helices (TMHMM): 0
⊟Accession numbers[edit | edit source]
⊟Protein sequence[edit | edit source]
- MNEEQRKASSVDVLAERDKKAEKDYSKYFEHVYQPPNLKEAKKRGKQEVRYNRDFQIDEKYRGMGNERTFLIKTYGCQMNAHDTEVIAGILEALGYQATTDINTADVILINTCAIRENAENKVFSEIGNLKHLKKERPDILIGVCGCMSQEESVVNKILKSYQNVDMIFGTHNIHHLPEILEEAYLSKAMVVEVWSKEGDVIENLPKVREGNIKAWVNIMYGCDKFCTYCIVPFTRGKERSRRPEDIIDEVRELAREGYKEITLLGQNVNSYGKDLQDIEYDLGDLLQAISKIAIPRVRFTTSHPWDFTDHMIDVISEGGNIVPHIHLPVQSGNNAVLKIMGRKYTRESYLDLVKRIKDRIPNVALTTDIIVGYPNESEEQFEETLTLYDEVGFEHAYTYLYSQRDGTPAAKMKDNVPLNVKKERLQRLNKKVGHYSQIAMSKYEGQTVTVLCEGSSKKDDQVLAGYTDKNKLVNFKAPKEMIGKLVEVRIDEAKQYSLNGSFIKEVEPEMVIQ
⊟Experimental data[edit | edit source]
- experimentally validated: PeptideAtlas [1] [2]
- protein localization: data available for COL
- quantitative data / protein copy number per cell: data available for COL
- interaction partners:
SAOUHSC_00530 elongation factor Tu [3] (data from MRSA252)
⊟Expression & Regulation[edit | edit source]
⊟Operon[edit | edit source]
- MicrobesOnline: SAOUHSC_01269 > SAOUHSC_01270 > SAOUHSC_01271predicted SigA promoter [4] : SAOUHSC_01266 > SAOUHSC_01267 > S529 > SAOUHSC_01268 > S530 > SAOUHSC_01269 > SAOUHSC_01270 > SAOUHSC_01271
⊟Regulation[edit | edit source]
- regulator:
⊟Transcription pattern[edit | edit source]
- S.aureus Expression Data Browser: [4]
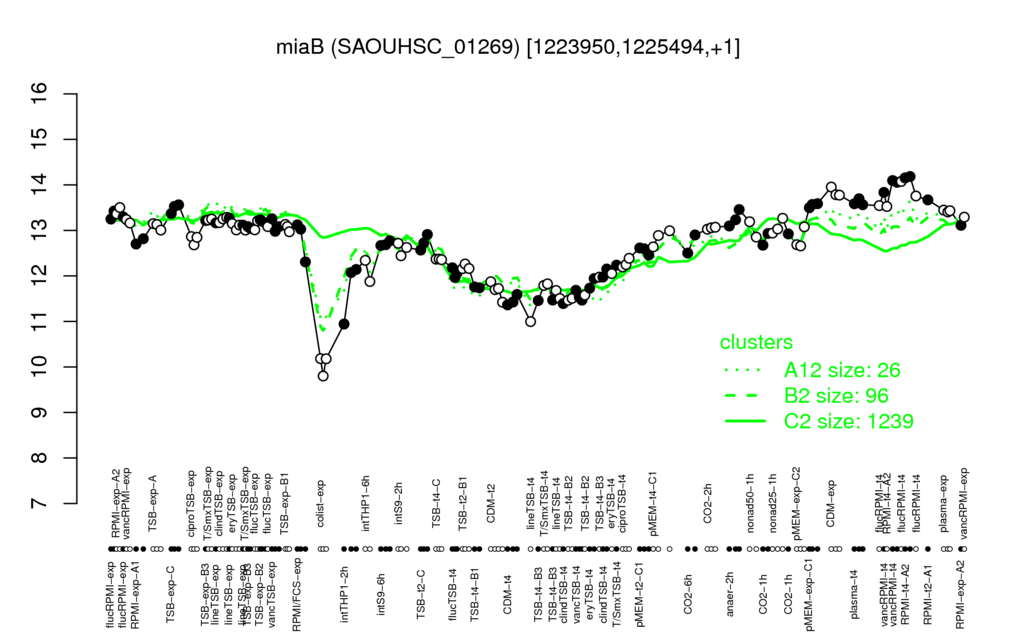 Multi-gene expression profiles
Multi-gene expression profiles
⊟Protein synthesis (provided by Aureolib)[edit | edit source]
- Aureolib: no data available
⊟Protein stability[edit | edit source]
- half-life: no data available
⊟Biological Material[edit | edit source]
⊟Mutants[edit | edit source]
⊟Expression vector[edit | edit source]
⊟lacZ fusion[edit | edit source]
⊟GFP fusion[edit | edit source]
⊟two-hybrid system[edit | edit source]
⊟FLAG-tag construct[edit | edit source]
⊟Antibody[edit | edit source]
⊟Other Information[edit | edit source]
You can add further information about the gene and protein here. [edit]
⊟Literature[edit | edit source]
⊟References[edit | edit source]
- ↑ Maren Depke, Stephan Michalik, Alexander Rabe, Kristin Surmann, Lars Brinkmann, Nico Jehmlich, Jörg Bernhardt, Michael Hecker, Bernd Wollscheid, Zhi Sun, Robert L Moritz, Uwe Völker, Frank Schmidt
A peptide resource for the analysis of Staphylococcus aureus in host-pathogen interaction studies.
Proteomics: 2015, 15(21);3648-61
[PubMed:26224020] [WorldCat.org] [DOI] (I p) - ↑ Stephan Michalik, Maren Depke, Annette Murr, Manuela Gesell Salazar, Ulrike Kusebauch, Zhi Sun, Tanja C Meyer, Kristin Surmann, Henrike Pförtner, Petra Hildebrandt, Stefan Weiss, Laura Marcela Palma Medina, Melanie Gutjahr, Elke Hammer, Dörte Becher, Thomas Pribyl, Sven Hammerschmidt, Eric W Deutsch, Samuel L Bader, Michael Hecker, Robert L Moritz, Ulrike Mäder, Uwe Völker, Frank Schmidt
A global Staphylococcus aureus proteome resource applied to the in vivo characterization of host-pathogen interactions.
Sci Rep: 2017, 7(1);9718
[PubMed:28887440] [WorldCat.org] [DOI] (I e) - ↑ Artem Cherkasov, Michael Hsing, Roya Zoraghi, Leonard J Foster, Raymond H See, Nikolay Stoynov, Jihong Jiang, Sukhbir Kaur, Tian Lian, Linda Jackson, Huansheng Gong, Rick Swayze, Emily Amandoron, Farhad Hormozdiari, Phuong Dao, Cenk Sahinalp, Osvaldo Santos-Filho, Peter Axerio-Cilies, Kendall Byler, William R McMaster, Robert C Brunham, B Brett Finlay, Neil E Reiner
Mapping the protein interaction network in methicillin-resistant Staphylococcus aureus.
J Proteome Res: 2011, 10(3);1139-50
[PubMed:21166474] [WorldCat.org] [DOI] (I p) - ↑ 4.0 4.1 4.2 Ulrike Mäder, Pierre Nicolas, Maren Depke, Jan Pané-Farré, Michel Debarbouille, Magdalena M van der Kooi-Pol, Cyprien Guérin, Sandra Dérozier, Aurelia Hiron, Hanne Jarmer, Aurélie Leduc, Stephan Michalik, Ewoud Reilman, Marc Schaffer, Frank Schmidt, Philippe Bessières, Philippe Noirot, Michael Hecker, Tarek Msadek, Uwe Völker, Jan Maarten van Dijl
Staphylococcus aureus Transcriptome Architecture: From Laboratory to Infection-Mimicking Conditions.
PLoS Genet: 2016, 12(4);e1005962
[PubMed:27035918] [WorldCat.org] [DOI] (I e)
