Jump to navigation
Jump to search
NCBI: 03-AUG-2016
⊟Summary[edit | edit source]
- organism: Staphylococcus aureus NCTC8325
- locus tag: SAOUHSC_01743
- pan locus tag?: SAUPAN004240000
- symbol: SAOUHSC_01743
- pan gene symbol?: apt
- synonym:
- product: adenine phosphoribosyltransferase
⊟Genome View[edit | edit source]
⊟Gene[edit | edit source]
⊟General[edit | edit source]
- type: CDS
- locus tag: SAOUHSC_01743
- symbol: SAOUHSC_01743
- product: adenine phosphoribosyltransferase
- replicon: chromosome
- strand: -
- coordinates: 1646766..1647284
- length: 519
- essential: no DEG other strains
⊟Accession numbers[edit | edit source]
- Gene ID: 3920543 NCBI
- RefSeq: YP_500250 NCBI
- BioCyc: G1I0R-1618 BioCyc
- MicrobesOnline: 1290164 MicrobesOnline
⊟Phenotype[edit | edit source]
Share your knowledge and add information here. [edit]
⊟DNA sequence[edit | edit source]
- 1
61
121
181
241
301
361
421
481ATGGATTTAAAGCAATACGTATCAGAAGTTCAAGATTGGCCGAAACCAGGTGTTAGTTTC
AAGGATATTACTACAATTATGGATAATGGTGAAGCATATGGCTATGCAACAGATAAAATT
GTAGAATACGCAAAAGACAGAGATGTTGATATCGTTGTAGGACCTGAAGCGCGTGGCTTT
ATCATTGGCTGTCCTGTAGCTTATTCAATGGGGATTGGCTTTGCACCTGTTAGAAAAGAA
GGGAAATTACCTCGTGAAGTCATTCGTTATGAGTATGACCTAGAATATGGTACAAATGTT
TTAACAATGCACAAAGATGCAATTAAACCAGGTCAACGTGTGTTAATTACAGATGATTTA
TTAGCTACTGGTGGTACGATTGAAGCAGCAATAAAATTAGTTGAAAAATTAGGCGGTATC
GTAGTAGGTATTGCATTTATAATTGAATTGAAATATTTAAATGGTATTGAAAAAATTAAA
GATTACGATGTTATGAGTTTAATCTCATACGACGAATAA60
120
180
240
300
360
420
480
519
⊟Protein[edit | edit source]
⊟General[edit | edit source]
- locus tag: SAOUHSC_01743
- symbol: SAOUHSC_01743
- description: adenine phosphoribosyltransferase
- length: 172
- theoretical pI: 4.48452
- theoretical MW: 19116.9
- GRAVY: -0.030814
⊟Function[edit | edit source]
- reaction: EC 2.4.2.7? ExPASyAdenine phosphoribosyltransferase AMP + diphosphate = adenine + 5-phospho-alpha-D-ribose 1-diphosphate
- TIGRFAM: Purines, pyrimidines, nucleosides, and nucleotides Salvage of nucleosides and nucleotides adenine phosphoribosyltransferase (TIGR01090; EC 2.4.2.7; HMM-score: 230.7)and 9 morePurines, pyrimidines, nucleosides, and nucleotides Purine ribonucleotide biosynthesis pur operon repressor PurR (TIGR01743; HMM-score: 47.6)Regulatory functions DNA interactions pur operon repressor PurR (TIGR01743; HMM-score: 47.6)Purines, pyrimidines, nucleosides, and nucleotides Pyrimidine ribonucleotide biosynthesis orotate phosphoribosyltransferase (TIGR01367; EC 2.4.2.10; HMM-score: 44.1)Purines, pyrimidines, nucleosides, and nucleotides Pyrimidine ribonucleotide biosynthesis orotate phosphoribosyltransferase (TIGR00336; EC 2.4.2.10; HMM-score: 37.2)Purines, pyrimidines, nucleosides, and nucleotides Salvage of nucleosides and nucleotides xanthine phosphoribosyltransferase (TIGR01744; EC 2.4.2.22; HMM-score: 35.5)Purines, pyrimidines, nucleosides, and nucleotides Purine ribonucleotide biosynthesis ribose-phosphate diphosphokinase (TIGR01251; EC 2.7.6.1; HMM-score: 27.9)Purines, pyrimidines, nucleosides, and nucleotides Salvage of nucleosides and nucleotides uracil phosphoribosyltransferase (TIGR01091; EC 2.4.2.9; HMM-score: 19.3)Purines, pyrimidines, nucleosides, and nucleotides Salvage of nucleosides and nucleotides hypoxanthine phosphoribosyltransferase (TIGR01203; EC 2.4.2.8; HMM-score: 14.6)Cellular processes DNA transformation comF family protein (TIGR00201; HMM-score: 13.3)
- TheSEED :
- Adenine phosphoribosyltransferase (EC 2.4.2.7)
Nucleosides and Nucleotides Purines Purine conversions Adenine phosphoribosyltransferase (EC 2.4.2.7)and 2 more - PFAM: PRTase-like (CL0533) Pribosyltran; Phosphoribosyl transferase domain (PF00156; HMM-score: 52.3)and 1 moreUPRTase; Uracil phosphoribosyltransferase (PF14681; HMM-score: 20)
⊟Structure, modifications & cofactors[edit | edit source]
- domains:
- modifications:
- cofactors:
- effectors:
⊟Localization[edit | edit source]
- PSORTb: Cytoplasmic
- Cytoplasmic Score: 10
- Cytoplasmic Membrane Score: 0
- Cellwall Score: 0
- Extracellular Score: 0
- Internal Helices: 0
- DeepLocPro: Cytoplasmic
- Cytoplasmic Score: 0.9589
- Cytoplasmic Membrane Score: 0.0015
- Cell wall & surface Score: 0.0002
- Extracellular Score: 0.0394
- LocateP: Intracellular
- Prediction by SwissProt Classification: Cytoplasmic
- Pathway Prediction: No pathway
- Intracellular possibility: 1
- Signal peptide possibility: -1
- N-terminally Anchored Score: 1
- Predicted Cleavage Site: No CleavageSite
- SignalP: no predicted signal peptide
- SP(Sec/SPI): 0.018018
- TAT(Tat/SPI): 0.000238
- LIPO(Sec/SPII): 0.001122
- predicted transmembrane helices (TMHMM): 0
⊟Accession numbers[edit | edit source]
⊟Protein sequence[edit | edit source]
- MDLKQYVSEVQDWPKPGVSFKDITTIMDNGEAYGYATDKIVEYAKDRDVDIVVGPEARGFIIGCPVAYSMGIGFAPVRKEGKLPREVIRYEYDLEYGTNVLTMHKDAIKPGQRVLITDDLLATGGTIEAAIKLVEKLGGIVVGIAFIIELKYLNGIEKIKDYDVMSLISYDE
⊟Experimental data[edit | edit source]
- experimentally validated: PeptideAtlas [1] [2]
- protein localization: data available for COL
- quantitative data / protein copy number per cell: data available for COL
- interaction partners:
SAOUHSC_01246 (infB) translation initiation factor IF-2 [3] (data from MRSA252) SAOUHSC_00943 (ppnK) inorganic polyphosphate/ATP-NAD kinase [3] (data from MRSA252) SAOUHSC_01232 (rpsB) 30S ribosomal protein S2 [3] (data from MRSA252) SAOUHSC_00336 acetyl-CoA acyltransferase [3] (data from MRSA252) SAOUHSC_00679 hypothetical protein [3] (data from MRSA252) SAOUHSC_01416 dihydrolipoamide succinyltransferase [3] (data from MRSA252)
⊟Expression & Regulation[edit | edit source]
⊟Operon[edit | edit source]
⊟Regulation[edit | edit source]
- regulator:
⊟Transcription pattern[edit | edit source]
- S.aureus Expression Data Browser: [4]
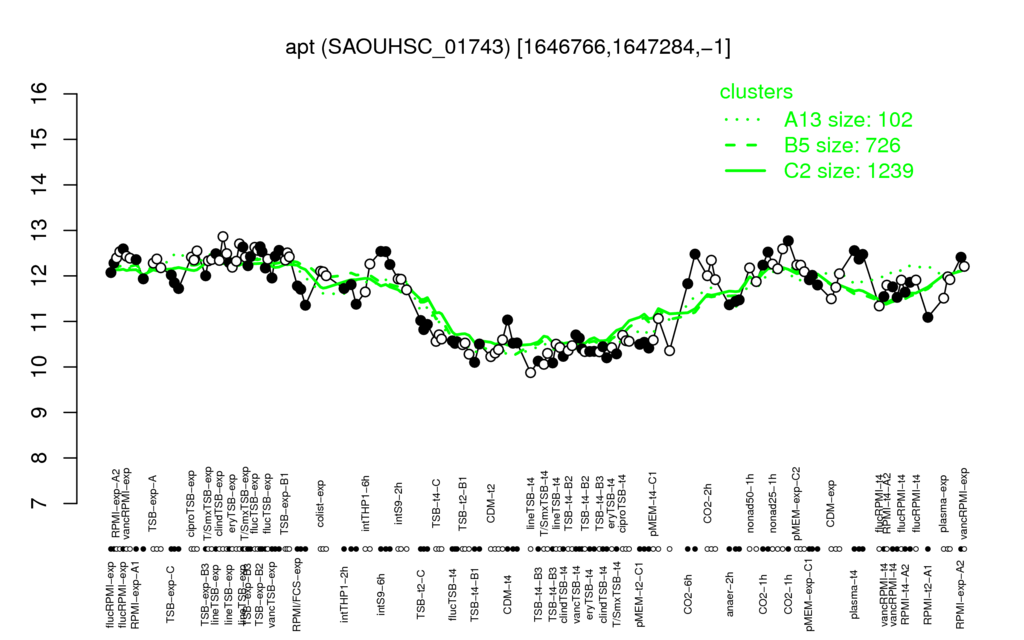 Multi-gene expression profiles
Multi-gene expression profiles
⊟Protein synthesis (provided by Aureolib)[edit | edit source]
- Aureolib: no data available
⊟Protein stability[edit | edit source]
- half-life: no data available
⊟Biological Material[edit | edit source]
⊟Mutants[edit | edit source]
⊟Expression vector[edit | edit source]
⊟lacZ fusion[edit | edit source]
⊟GFP fusion[edit | edit source]
⊟two-hybrid system[edit | edit source]
⊟FLAG-tag construct[edit | edit source]
⊟Antibody[edit | edit source]
⊟Other Information[edit | edit source]
You can add further information about the gene and protein here. [edit]
⊟Literature[edit | edit source]
⊟References[edit | edit source]
- ↑ Maren Depke, Stephan Michalik, Alexander Rabe, Kristin Surmann, Lars Brinkmann, Nico Jehmlich, Jörg Bernhardt, Michael Hecker, Bernd Wollscheid, Zhi Sun, Robert L Moritz, Uwe Völker, Frank Schmidt
A peptide resource for the analysis of Staphylococcus aureus in host-pathogen interaction studies.
Proteomics: 2015, 15(21);3648-61
[PubMed:26224020] [WorldCat.org] [DOI] (I p) - ↑ Stephan Michalik, Maren Depke, Annette Murr, Manuela Gesell Salazar, Ulrike Kusebauch, Zhi Sun, Tanja C Meyer, Kristin Surmann, Henrike Pförtner, Petra Hildebrandt, Stefan Weiss, Laura Marcela Palma Medina, Melanie Gutjahr, Elke Hammer, Dörte Becher, Thomas Pribyl, Sven Hammerschmidt, Eric W Deutsch, Samuel L Bader, Michael Hecker, Robert L Moritz, Ulrike Mäder, Uwe Völker, Frank Schmidt
A global Staphylococcus aureus proteome resource applied to the in vivo characterization of host-pathogen interactions.
Sci Rep: 2017, 7(1);9718
[PubMed:28887440] [WorldCat.org] [DOI] (I e) - ↑ 3.0 3.1 3.2 3.3 3.4 3.5 Artem Cherkasov, Michael Hsing, Roya Zoraghi, Leonard J Foster, Raymond H See, Nikolay Stoynov, Jihong Jiang, Sukhbir Kaur, Tian Lian, Linda Jackson, Huansheng Gong, Rick Swayze, Emily Amandoron, Farhad Hormozdiari, Phuong Dao, Cenk Sahinalp, Osvaldo Santos-Filho, Peter Axerio-Cilies, Kendall Byler, William R McMaster, Robert C Brunham, B Brett Finlay, Neil E Reiner
Mapping the protein interaction network in methicillin-resistant Staphylococcus aureus.
J Proteome Res: 2011, 10(3);1139-50
[PubMed:21166474] [WorldCat.org] [DOI] (I p) - ↑ 4.0 4.1 Ulrike Mäder, Pierre Nicolas, Maren Depke, Jan Pané-Farré, Michel Debarbouille, Magdalena M van der Kooi-Pol, Cyprien Guérin, Sandra Dérozier, Aurelia Hiron, Hanne Jarmer, Aurélie Leduc, Stephan Michalik, Ewoud Reilman, Marc Schaffer, Frank Schmidt, Philippe Bessières, Philippe Noirot, Michael Hecker, Tarek Msadek, Uwe Völker, Jan Maarten van Dijl
Staphylococcus aureus Transcriptome Architecture: From Laboratory to Infection-Mimicking Conditions.
PLoS Genet: 2016, 12(4);e1005962
[PubMed:27035918] [WorldCat.org] [DOI] (I e)
