Jump to navigation
Jump to search
NCBI: 10-JUN-2013
⊟Summary[edit | edit source]
- organism: Staphylococcus aureus COL
- locus tag: SACOL0877 [new locus tag: SACOL_RS04500 ]
- pan locus tag?: SAUPAN002821000
- symbol: gcvH
- pan gene symbol?: gcvH
- synonym:
- product: glycine cleavage system protein H
⊟Genome View[edit | edit source]
⊟Gene[edit | edit source]
⊟General[edit | edit source]
- type: CDS
- locus tag: SACOL0877 [new locus tag: SACOL_RS04500 ]
- symbol: gcvH
- product: glycine cleavage system protein H
- replicon: chromosome
- strand: +
- coordinates: 897692..898072
- length: 381
- essential: unknown other strains
⊟Accession numbers[edit | edit source]
- Gene ID: 3238420 NCBI
- RefSeq: YP_185749 NCBI
- BioCyc: see SACOL_RS04500
- MicrobesOnline: 912349 MicrobesOnline
⊟Phenotype[edit | edit source]
Share your knowledge and add information here. [edit]
⊟DNA sequence[edit | edit source]
- 1
61
121
181
241
301
361TTGGCAGTACCAAATGAATTGAAATATTCAAAAGAGCATGAATGGGTTAAAGTTGAAGGT
AATGTAGCAATAATTGGAATCACAGAATACGCACAAAGCGAGTTAGGTGATATTGTTTTC
GTTGAATTACCAGAAACAGATGATGAAATTAATGAAGGGGATACGTTTGGTAGCGTAGAA
TCAGTTAAAACTGTATCAGAATTATATGCACCAATCTCTGGTAAAGTAGTTGAAGTCAAC
GAAGAACTAGAAGATAGTCCCGAATTTGTAAATGAATCTCCATACGAAAAAGCATGGATG
GTAAAAGTAGAAATTAGTGATGAAAGTCAGATTGAAGCTTTATTAACAGCTGAAAAATAT
TCAGAAATGATTGGTGAATAA60
120
180
240
300
360
381
⊟Protein[edit | edit source]
⊟General[edit | edit source]
- locus tag: SACOL0877 [new locus tag: SACOL_RS04500 ]
- symbol: GcvH
- description: glycine cleavage system protein H
- length: 126
- theoretical pI: 3.70004
- theoretical MW: 14092.5
- GRAVY: -0.333333
⊟Function[edit | edit source]
- TIGRFAM: Energy metabolism Amino acids and amines glycine cleavage system H protein (TIGR00527; HMM-score: 184.5)and 3 moreglycine cleavage protein H-like protein (TIGR03077; HMM-score: 69.5)Fatty acid and phospholipid metabolism Biosynthesis acetyl-CoA carboxylase, biotin carboxyl carrier protein (TIGR00531; HMM-score: 15.2)Central intermediary metabolism Nitrogen metabolism urea carboxylase (TIGR02712; EC 6.3.4.6; HMM-score: 11.1)
- TheSEED :
- Glycine cleavage system H protein
Amino Acids and Derivatives Alanine, serine, and glycine Glycine and Serine Utilization Glycine cleavage system H proteinand 1 more - PFAM: Hybrid (CL0105) GCV_H; Glycine cleavage H-protein (PF01597; HMM-score: 165.4)and 2 moreBiotin_lipoyl; Biotin-requiring enzyme (PF00364; HMM-score: 16.2)RnfC_N; RnfC Barrel sandwich hybrid domain (PF13375; HMM-score: 15.2)
⊟Structure, modifications & cofactors[edit | edit source]
- domains:
- modifications:
- cofactors: (R)-lipoate
- effectors:
⊟Localization[edit | edit source]
- PSORTb: Cytoplasmic
- Cytoplasmic Score: 7.5
- Cytoplasmic Membrane Score: 1.15
- Cellwall Score: 0.62
- Extracellular Score: 0.73
- Internal Helices: 0
- DeepLocPro: Cytoplasmic
- Cytoplasmic Score: 0.996
- Cytoplasmic Membrane Score: 0.0004
- Cell wall & surface Score: 0
- Extracellular Score: 0.0036
- LocateP: Intracellular
- Prediction by SwissProt Classification: Cytoplasmic
- Pathway Prediction: No pathway
- Intracellular possibility: 1
- Signal peptide possibility: -1
- N-terminally Anchored Score: 1
- Predicted Cleavage Site: No CleavageSite
- SignalP: no predicted signal peptide
- SP(Sec/SPI): 0.003128
- TAT(Tat/SPI): 0.000167
- LIPO(Sec/SPII): 0.000284
- predicted transmembrane helices (TMHMM): 0
⊟Accession numbers[edit | edit source]
⊟Protein sequence[edit | edit source]
- MAVPNELKYSKEHEWVKVEGNVAIIGITEYAQSELGDIVFVELPETDDEINEGDTFGSVESVKTVSELYAPISGKVVEVNEELEDSPEFVNESPYEKAWMVKVEISDESQIEALLTAEKYSEMIGE
⊟Experimental data[edit | edit source]
- experimentally validated: PeptideAtlas
- protein localization: Cytoplasmic [1] [2] [3]
- quantitative data / protein copy number per cell: 2826 [4]
- interaction partners:
SACOL2656 (arcB2) ornithine carbamoyltransferase [5] (data from MRSA252) SACOL1595 (gcvT) glycine cleavage system aminomethyltransferase T [5] (data from MRSA252) SACOL1513 (hup) DNA-binding protein HU [5] (data from MRSA252) SACOL1741 (icd) isocitrate dehydrogenase [5] (data from MRSA252) SACOL1102 (pdhA) pyruvate dehydrogenase complex E1 component subunit alpha [5] (data from MRSA252) SACOL1103 (pdhB) pyruvate dehydrogenase complex E1 component subunit beta [5] (data from MRSA252) SACOL1104 (pdhC) branched-chain alpha-keto acid dehydrogenase E2 [5] (data from MRSA252) SACOL1105 (pdhD) dihydrolipoamide dehydrogenase [5] (data from MRSA252) SACOL0584 (rplA) 50S ribosomal protein L1 [5] (data from MRSA252) SACOL2236 (rplB) 50S ribosomal protein L2 [5] (data from MRSA252) SACOL2239 (rplC) 50S ribosomal protein L3 [5] (data from MRSA252) SACOL2227 (rplE) 50S ribosomal protein L5 [5] (data from MRSA252) SACOL2220 (rplO) 50S ribosomal protein L15 [5] (data from MRSA252) SACOL1257 (rplS) 50S ribosomal protein L19 [5] (data from MRSA252) SACOL1725 (rplT) 50S ribosomal protein L20 [5] (data from MRSA252) SACOL2234 (rplV) 50S ribosomal protein L22 [5] (data from MRSA252) SACOL1726 (rpmI) 50S ribosomal protein L35 [5] (data from MRSA252) SACOL2222 (rpsE) 30S ribosomal protein S5 [5] (data from MRSA252) SACOL0592 (rpsG) 30S ribosomal protein S7 [5] (data from MRSA252) SACOL2215 (rpsM) 30S ribosomal protein S13 [5] (data from MRSA252) SACOL2235 (rpsS) 30S ribosomal protein S19 [5] (data from MRSA252) SACOL1449 (sucA) 2-oxoglutarate dehydrogenase E1 component [5] (data from MRSA252) SACOL1448 (sucB) dihydrolipoamide succinyltransferase [5] (data from MRSA252) SACOL0594 (tuf) elongation factor Tu [5] (data from MRSA252) SACOL1020 hypothetical protein [5] (data from MRSA252) SACOL1593 glycine dehydrogenase subunit 2 [5] (data from MRSA252) SACOL1594 glycine dehydrogenase subunit 1 [5] (data from MRSA252)
⊟Expression & Regulation[edit | edit source]
⊟Operon[edit | edit source]
- MicrobesOnline: no polycistronic organisation predicted
⊟Regulation[edit | edit source]
- regulator: CcpA regulon
CcpA (TF) important in Carbon catabolism; RegPrecise
⊟Transcription pattern[edit | edit source]
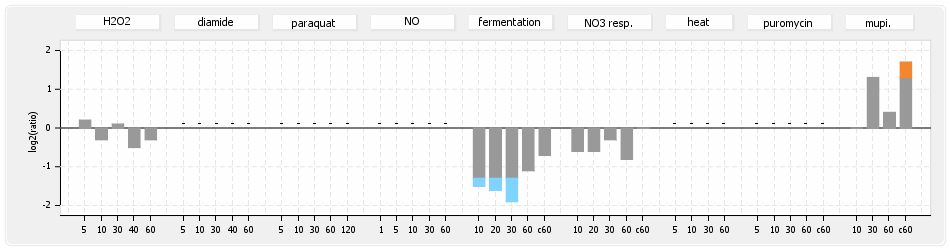
- S.aureus Expression Data Browser: data available for NCTC8325
⊟Protein synthesis (provided by Aureolib)[edit | edit source]
⊟Protein stability[edit | edit source]
- half-life: no data available
⊟Biological Material[edit | edit source]
⊟Mutants[edit | edit source]
⊟Expression vector[edit | edit source]
⊟lacZ fusion[edit | edit source]
⊟GFP fusion[edit | edit source]
⊟two-hybrid system[edit | edit source]
⊟FLAG-tag construct[edit | edit source]
⊟Antibody[edit | edit source]
⊟Other Information[edit | edit source]
You can add further information about the gene and protein here. [edit]
⊟Literature[edit | edit source]
⊟References[edit | edit source]
- ↑ Dörte Becher, Kristina Hempel, Susanne Sievers, Daniela Zühlke, Jan Pané-Farré, Andreas Otto, Stephan Fuchs, Dirk Albrecht, Jörg Bernhardt, Susanne Engelmann, Uwe Völker, Jan Maarten van Dijl, Michael Hecker
A proteomic view of an important human pathogen--towards the quantification of the entire Staphylococcus aureus proteome.
PLoS One: 2009, 4(12);e8176
[PubMed:19997597] [WorldCat.org] [DOI] (I e) - ↑ Kristina Hempel, Florian-Alexander Herbst, Martin Moche, Michael Hecker, Dörte Becher
Quantitative proteomic view on secreted, cell surface-associated, and cytoplasmic proteins of the methicillin-resistant human pathogen Staphylococcus aureus under iron-limited conditions.
J Proteome Res: 2011, 10(4);1657-66
[PubMed:21323324] [WorldCat.org] [DOI] (I p) - ↑ Andreas Otto, Jan Maarten van Dijl, Michael Hecker, Dörte Becher
The Staphylococcus aureus proteome.
Int J Med Microbiol: 2014, 304(2);110-20
[PubMed:24439828] [WorldCat.org] [DOI] (I p) - ↑ Daniela Zühlke, Kirsten Dörries, Jörg Bernhardt, Sandra Maaß, Jan Muntel, Volkmar Liebscher, Jan Pané-Farré, Katharina Riedel, Michael Lalk, Uwe Völker, Susanne Engelmann, Dörte Becher, Stephan Fuchs, Michael Hecker
Costs of life - Dynamics of the protein inventory of Staphylococcus aureus during anaerobiosis.
Sci Rep: 2016, 6;28172
[PubMed:27344979] [WorldCat.org] [DOI] (I e) - ↑ 5.00 5.01 5.02 5.03 5.04 5.05 5.06 5.07 5.08 5.09 5.10 5.11 5.12 5.13 5.14 5.15 5.16 5.17 5.18 5.19 5.20 5.21 5.22 5.23 5.24 5.25 5.26 Artem Cherkasov, Michael Hsing, Roya Zoraghi, Leonard J Foster, Raymond H See, Nikolay Stoynov, Jihong Jiang, Sukhbir Kaur, Tian Lian, Linda Jackson, Huansheng Gong, Rick Swayze, Emily Amandoron, Farhad Hormozdiari, Phuong Dao, Cenk Sahinalp, Osvaldo Santos-Filho, Peter Axerio-Cilies, Kendall Byler, William R McMaster, Robert C Brunham, B Brett Finlay, Neil E Reiner
Mapping the protein interaction network in methicillin-resistant Staphylococcus aureus.
J Proteome Res: 2011, 10(3);1139-50
[PubMed:21166474] [WorldCat.org] [DOI] (I p)